Pediatrics International © 2011 Japan Pediatric Society. The new glucometer (Nova-Statstrip(®) ) could be used for point-of-care blood glucose measurement in neonates as it showed a narrow margin of error and had no hematocrit or bilirubin interference. In terms of test repeatability, performance results for the ED physician were not comparable to that of the laboratory technologist. The test for glucose, as part of the i-STAT System, is intended for use in the in. Arterial or venous: syringe or tube with lithium heparin. With the iSTAT 1 System, the FDA has categorized the Glucose Cartridge as a Non-Waived Complex Test when testing is performed using arterial, venous, and capillary whole blood samples. Acceptable Samples for PCx and PCx Plus Glucose Test Strips. 
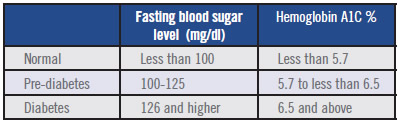
No significant interference of hematocrit or total serum bilirubin was found on the mean bias of the Statstrip(®). averaging -18.3 mmHg with a wide range from -84.6 to 48.1 mmHg. The iSTAT 1 analyzer is intended for use in approved Point of Care locations. At a hypoglycemic level (<45 mg/dL), it showed a sensitivity of 95.2%. The mean difference (☒SD) of the Statstrip(®) was 2.8 (-14.1, 19.7) mg/dL. Clinical Chemistry Reference Intervals Veterinary Medical Teaching Hospital University of California, Davis Analyte Unit Dog Cat Horse Eq. At a glucose concentration more than 75 mg/dL, 97% of the Statstrip(®) readings were within 20% of the reference values. At plasma glucose concentrations less than 75 mg/dL, the Statstrip(®) achieved 93% in the tests for discrepancy < 15 mg/dL. Twenty-one specimens had plasma glucose concentrations <45 mg/dL. In the normal pO 2 range (10.6 < pO 2 <13.3 kPa), the performance of the i-STAT was comparable to the RapidLab. E 6.2 Assess glycemic status at least quarterly, and as needed, in patients whose therapy has recently changed and/or who are not meeting glycemic goals. One hundred and fifty-one blood specimens were collected and measured by the reference method with plasma glucose concentrations ranging from 12 to 371 mg/dL. Recommendations 6.1 Assess glycemic status (A1C or other glycemic measurement) at least two times a year in patients who are meeting treatment goals (and who have stable glycemic control). Hematocrit and total serum bilirubin measurements were performed simultaneously. Standard reference was performed using the hexokinase method within 10 min of blood collection. Venous blood specimens of neonates were collected and tested by the two glucometers. The aim of this study was to evaluate the performance of the new glucometer (Nova-Statstrip(®) Nova Biomedical, Waltham, MA, USA) compared to the reference method. Performance of point-of-care (POC) glucometers in newborns have been unsatisfactory in low glucose concentration range and the effects of different hematocrit levels on glucose measurements have also demonstrated in currently used POC glucometers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
